|
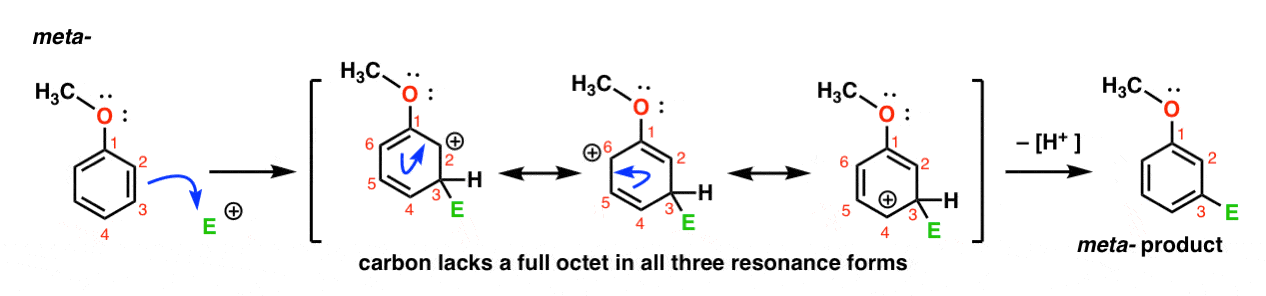
explain why the electrophilic aromatic substitution of halobenzenes proceeds more slowly than does the electrophilic substitution of benzene itself.explain, by drawing the resonance contributors for the intermediate carbocation, why the electrophilic aromatic substitution of halobenzenes produces a mixture of mainly ortho- and para-substituted products. Disubstituted benzenes When there are two (or more) substituents, the relative position of the subsituents must be defined.explain why electrophilic aromatic substitution of benzene derivatives containing one of the substituents listed in Objective 7, above, proceeds more slowly than the electrophilic substitution of benzene itself.explain, by drawing the resonance contributors for the intermediate carbocation, why meta substitution predominates in electrophilic aromatic substitution reactions carried out on benzene derivatives containing one of the substituents R 3N +, NO 2, CO 2H, CN, CO 2R, COR or CHO.explain why the electrophilic substitution of phenols, amines and their derivatives proceeds more rapidly than the electrophilic substitution of benzene itself. 
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
