|
Here, we review data on the carcinogenic mechanisms of Hsp90 and their inhibitors. This, in turn, will reveal clues for developing specific treatment centered on the chaperone, for instance by inhibiting its pro-carcinogenic functions (negative chaperonotherapy). 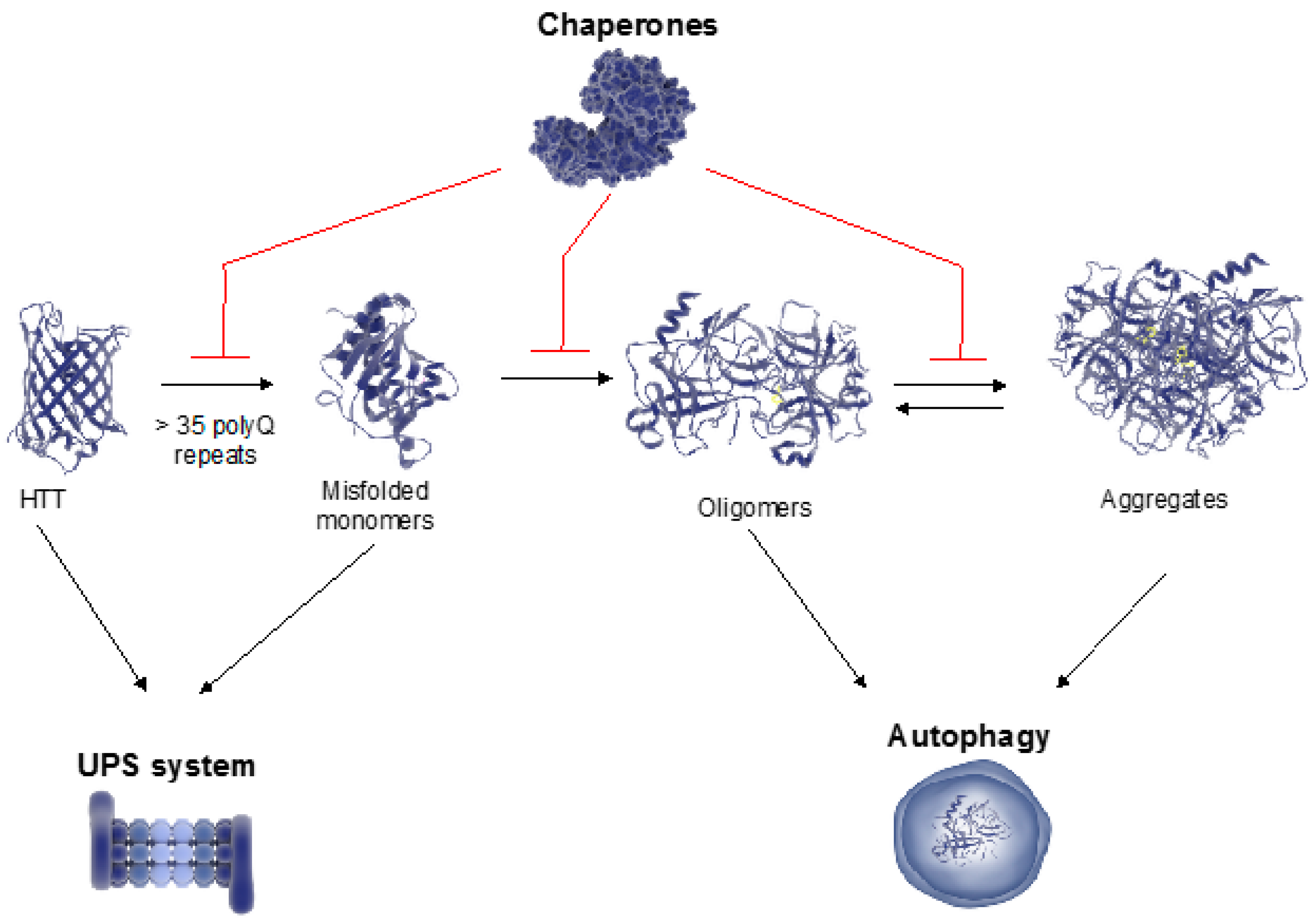
Quantitative data available on this chaperone in salivary gland tissue with inflammation, and benign and malignant tumors suggest that assessing tissue Hsp90 levels and distribution patterns is useful for differential diagnosis-prognostication, and patient follow up. Some chaperones such as Hsp90 potentiate tumor growth, proliferation, and metastasization. Chaperones are cytoprotective, but can also be etiopathogenic agents causing diseases, the chaperonopathies. Previous studies pertaining to the CS of the salivary glands have determined the quantitative and distribution patterns for several members, the chaperones, in normal and diseased glands, focusing on tumors. It is present throughout the body but with distinctive features for each cell and tissue type. Research from this dissertation will contribute to further understanding Alzheimer’s disease pathogenesis, and, more broadly, the breakdown of protein homeostasis in neurodegenerative disease, and roles for chaperone proteins in managing proteotoxic aggregates in possible future therapies.The chaperone system (CS) of an organism is composed of molecular chaperones, chaperone co-factors, co-chaperones, and chaperone receptors and interactors. Lastly, in an Alzheimer’s disease model of hyperphosphylated tau, we monitored changes in chaperone protein levels in response to inhibition of protein phosphatases. We observed that ClpX can bind and reactivate native and engineered protein aggregates in the absence of ATP. Moreover, we identified a novel function of the chaperone protein ClpX in protein disaggregation in vitro and in vivo. Furthermore, we investigated the mechanism of substrate recognition and amyloid disassembly by Hsp104 in a yeast prion model of amyloid assembly. We studied the role of molecular chaperone proteins in the progression of protein aggregation in a model of Alzheimer’s disease and developed novel chaperone tools for targeting amyloid proteins for protein clearance. Here, we investigated the biochemical properties of the Hsp100/Clp chaperone protein family, which couples ATP binding and hydrolysis to unfold and reactive aggregated and misfolded polypeptides. This thesis investigates the role of ATP-dependent chaperone proteins in disaggregating and resolubilizing protein aggregates, including model aggregates, amyloids of Sup35 in yeast and hyperphosphylated tau in human cells. It is thought that protein aggregation in neurodegenerative diseases results from disturbances in pathways that regulate protein quality control. Given that these diseases share a common theme of protein misfolding and aggregation, researchers have questioned whether molecular chaperone proteins are involved in disease pathology, which has led to investigations into the possible use of chaperone-based strategies as treatment options. In each of these protein misfolding diseases, the roles of chaperone proteins are complex and not well understood. The neurodegenerative diseases Alzheimer’s disease, Huntington’s disease, and Parkinson’s disease are characterized by protein misfolding and accumulation into aggregates composed of amyloid fibrils. It is thought that neurodegenerative diseases may be the result of a derailed proteostasis network in response to aging, mutations, and environmental stress, among other factors that contribute to protein aggregation. Moreover, molecular chaperones are required for folding denatured, misfolded, and de novo proteins into their native conformation. 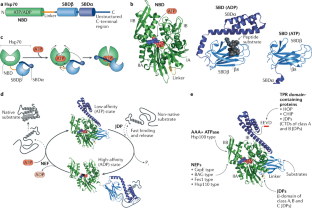
Molecular chaperone proteins function in various cellular processes including intracellular transport, oligomeric assembly, and efficient protein folding. This network is referred to as the proteostasis network and includes molecular chaperone proteins, co-chaperone proteins, and the ubiquitin proteasome system. Within the cell, a robust network of proteins safeguard against protein misfolding and aggregation. The exposed regions of two or more proteins associate to form amorphous deposits or highly ordered, stable fibrillar structures called amyloid aggregates. Protein aggregation occurs when proteins adopt non-native conformations, exposing hydrophobic surfaces due to misfolding.
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
